

Bivalves are exploited because they are very productive, low in the food chain, abundant in shallow coastal waters and are tasty and nutritious. World production of bivalves from both fisheries and aquaculture is dominated by the exploitation of mussels, oysters, scallops and clams for food. Indeed, over-fishing of some stocks of these taxa has driven the development of bivalve husbandry and aquaculture for centuries. In addition, bivalves are exploited for pearls and the shell as well as the soft edible tissues and these 'exotic' products are the basis of multi-million dollar exports from some parts of the world. Demand for these products has also led to overfishing which resulted in the development of highly specialist pearl-culture technologies and the aquaculture of bivalves for conservation and restoration.
 The
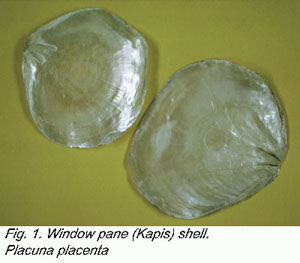
window pane shell Placuna placenta (Linnaeus, 1758) (Fig. 1)
is a bivalve with a very translucent shell that was used traditionally
in windows in Asia and, more recently, for shell-craft items (Fig.
3). It also produces pearls that are exploited commercially (Dharmaraj
et al., 2004). The species has a wide distribution including
the Gulf of Aden and around India, the Malay Peninsula, the southern
coasts of China and along the northern coasts of Borneo to the Philippines
(Yonge, 1977). Four congeneric species are currently recognised in
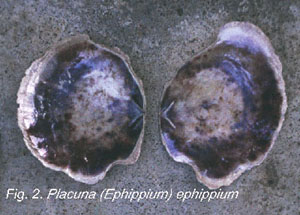
the Family Placunidae: Placuna placenta; P. (Ephippium)
lincolnii (Gray, 1849); P. (Ephippium) ephippium
(Philipsson, 1788) (Fig. 2);and . (Ephippium) lobata Sowerby,1871
(http://www.data.acnatsci.org/obis/)
but only P. placenta has a shell that is sufficiently translucent
for commercial use.
The
window pane shell Placuna placenta (Linnaeus, 1758) (Fig. 1)
is a bivalve with a very translucent shell that was used traditionally
in windows in Asia and, more recently, for shell-craft items (Fig.
3). It also produces pearls that are exploited commercially (Dharmaraj
et al., 2004). The species has a wide distribution including
the Gulf of Aden and around India, the Malay Peninsula, the southern
coasts of China and along the northern coasts of Borneo to the Philippines
(Yonge, 1977). Four congeneric species are currently recognised in
the Family Placunidae: Placuna placenta; P. (Ephippium)
lincolnii (Gray, 1849); P. (Ephippium) ephippium
(Philipsson, 1788) (Fig. 2);and . (Ephippium) lobata Sowerby,1871
(http://www.data.acnatsci.org/obis/)
but only P. placenta has a shell that is sufficiently translucent
for commercial use.
Biology
 Placuna
placenta is found on muddy or sandy substrata from shallow water
to depths of up to 100m. Kapis shells can be found in bays, coves
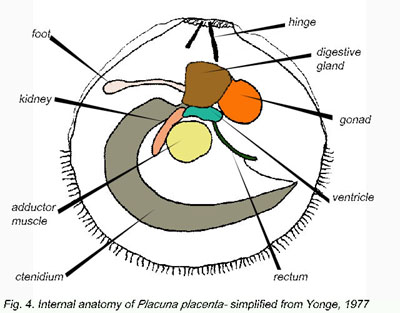
and estuaries. Form and structure and the organs of the mantle cavity
(Fig. 4), which are typical of most monomyarian bivalves, are described
in detail in Yonge (1977). The shell is extremely flattened laterally,
the valves are thin with a V-shaped ligament (Figs. 1, 4 ) and can
exceed 150 mm in length. Like other bivalves, Window-pane shells are
filter feeders. When in water, they open their shells a little. A
current of water is generated through their shells and they sieve
out the food particles with their enlarged gills. When exposed at
low tide, the valves are clamped closed. Their diets consist primarily
of plankton and organic detritus. The sexes are separate, and the
males and females are easily differentiated externally by colour of
the gonad. Individuals mature at between 70 and 100 mm and fertilization
is external. Larval development is described in Young (1980). Larvae
are planktonic for around 14 days and probably attach byssally at
metamorphosis and newly settled juveniles can burrow partially into
mud (Young, 1980).
Placuna
placenta is found on muddy or sandy substrata from shallow water
to depths of up to 100m. Kapis shells can be found in bays, coves
and estuaries. Form and structure and the organs of the mantle cavity
(Fig. 4), which are typical of most monomyarian bivalves, are described
in detail in Yonge (1977). The shell is extremely flattened laterally,
the valves are thin with a V-shaped ligament (Figs. 1, 4 ) and can
exceed 150 mm in length. Like other bivalves, Window-pane shells are
filter feeders. When in water, they open their shells a little. A
current of water is generated through their shells and they sieve
out the food particles with their enlarged gills. When exposed at
low tide, the valves are clamped closed. Their diets consist primarily
of plankton and organic detritus. The sexes are separate, and the
males and females are easily differentiated externally by colour of
the gonad. Individuals mature at between 70 and 100 mm and fertilization
is external. Larval development is described in Young (1980). Larvae
are planktonic for around 14 days and probably attach byssally at
metamorphosis and newly settled juveniles can burrow partially into
mud (Young, 1980).
 Economic
Importance
Economic
Importance
The meat is eaten but most of Placuna's value comes from its shells which are used in animal glue, chalk, shellac, soldering lead and paint as well as decoratively. Although window-pane shells are harvested in other parts of the world such as India (Dharmaraj et al., 2004) the major producing country is the Philippines which exported US$36 million worth of Kapis products between 1986 and 1991 (Gallardo et al., 1995). Major sources in the Philippines are listed by Gallardo et al. (1995). In 1991, Kapis was ranked fifth after prawns, tuna, seaweeds and octopus among the major fishery exports of the Philippines (Philippine Fisheries Profile 1991). Major markets include U.S.A, Japan, Germany and the rest of Europe. Kapis shells are gathered from the wild by hand, diving and dredging and graded according to size and quality. Shells that are bigger than 75 mm are classified as first class while those less than 60 mm are fourth class. To reduce damage to the shells, they are allowed to dry and open naturally. The meat is then removed and, because of its high protein content, is often used as a component for poultry and shrimp feeds.
A decline in Kapis shell production in the Philippines has been observed since the 1970s. Between 1994 and 1999 Kapis exports declined from 3260 MT to 1765 MT (Madrones-Ladja, 2002). The main cause of the decline in the Philippines and elsewhere is over exploitation exacerbated by destructive methods of fishing and gathering such as trawling, using mechanical rakes and dredges, dynamite fishing and compressor diving. Destructive fishing for kapis kills recruits and brood stock and destroys the bottom substrate.It has also been suggested that the increase in prawn hatcheries is also responsible for the decline of Kapis. In the late 1980s prawn hatcheries flushed water laced with antibiotics back to the sea and the contamination may have killed Kapis. Attempts to slow the decline in this valuable resource include fisheries regulations to control harvesting and aquaculture (Madrones-Ladja, 2002; Dharmaraj et al. 2004). In the Philippines, fisheries regulations involves issuing licences and permits, setting minimum and maximum landing sizes and establishing restricted areas (http://www.bfar.da.gov.ph/legislation/fao/fao157.htm). Regulations are supported by fines and the threat of imprisonment. However, the high market demand means gatherers continue to collect all they can even though the government prohibits the harvest of shells with sizes less than 80 mm and more than 100 mm, and natural resources continue to be depleted.
 Cultivation
Cultivation
Placuna was identified as the second priority mollusc species for research during the Second Conference on Aquaculture Development in Southeast Asia (ADSEA) held in the Philippines in 1994 (Madrones-Ladja, 1997). Attempts have been made to artificially propagate this species in the Philippines and in India because of the very wide fluctuations in natural recruitment from year to year. Ripe broodstock are taken from the wild and induced to spawn using photochemical (Madrones-Ladja, 1997) or thermal (Dharmaraj et al., 2004) stimulation. Straight-hinged larvae are formed within twenty-four hours after fertilisation and are sieved, transferred to rearing containers and fed on unicellular microalgae such as Isochrysis galbana. Under optimal conditions settlement and metamorphosis occurs after about 14 days at 24-27°C but is delayed at elevated or reduced salinities (Madrones et al., 2002). Spat settlement may occur in a shorter time at higher temperatures (8 days at 28-30°C) (Dharmaraj et al., 2004). To maximise broodstock fecundity while maintaining egg quality and larval viability, the effectiveness of gonad conditioning methods has also been evaluated. Gonad development of P. placenta can be hastened by providing a suitable micro algal diet and rearing conditions (Madrones et.al., 2002).
Restocking
Conservation of Placuna placenta currently involves transplantation of wild-caught juveniles and broodstock from areas where stocks are abundant to depleted areas with suitable ground. Kapis shells can be grown in areas with water temperature 24.5-30oC, salinity 18-38 ppt, pH 6.4-7.7 and dissolved oxygen 2.5-5 ppm. Ideally in transplanted areas density should be limited to approximately 150-200 per m2 (1.5-2.0 million seedlings per hectare) to allow normal growth and prevent overcrowding.
The Southeast Asian Fisheries Development Centre - Aquaculture Department (SEAFDEC-AQD) started research in 1990 on the feasibility of restocking a depleted kapis bed in Panay Gulf, Phillippines, with stock from an abundant area. (http://www.seafdec.org.ph/stories/story06-stock-enhancement-program.html). Panay Gulf still is a suitable ground for kapis beds. The restocking was an initial success with juveniles present the following year. However local cooperation was limited and gatherers collected the juveniles, thus reducing the number left to breed successive generations. There were also illegal trawls and dredges in the project area. Future work will necessitate refinement of brood-stock conditioning and development of techniques for rearing larvae, intermediate culture and grow-out.
The way forward for the human use of this species is through sustainable harvesting. To do this local education programmes are needed and the local communities will have to be involved. Traders of Kapis shell are also becoming more aware of how limited the resource is with items advertised as being harvested sustainably, 'ensuring that future generations will be able to bask in the glow of light transmitted through this remarkable material' (Edzina web site).Claire Campbell, School of Biology and Biochemistry, Queen's University, Belfast.
References
| Dharmaraj S, Shanmugasundaraman K & Suja C P (2004) Larval rearing and spat production |
|
| of the windowpane shell Placuna placenta. Aquaculture Asia, 9: 20-28. | |
| Gallardo W G, Siar S V & Encena V (1995) Exploitation of the Window Pane Shell Placuna |
|
| placenta in the Philippines. Biological Conservation, 73: 33-38. | |
| Madrones-Ladja J A (1997) Notes on the induced spawning, embryonic and larval |
|
| development of the window pane-shell, Placuna placenta (Linnaeus, 1758), in the | |
| laboratory. Aquaculture, 157: 137-146. | |
| Madrones-Ladja J A (2002) Salinity effect on the embryonic development, larval growth |
|
| and survival at metamorphosis of Placuna placenta Linnaeus (1758). Aquaculture, | |
| 214: 411-418. | |
| Madrones-Ladja J A, de la Pena M R & Parami N P (2002) The effect of micro algal |
|
| diet and rearing condition on gonad maturity, fecundity and embryonic development of the | |
| window pane shell, Placuna placenta Linnaeus. Aquaculture, 206: 313-321. | |
| Yonge C M (1977) Form and evolution in the Anomiacea (Mollusca: Bivalvia) - Pododesmus, |
|
| Anomia, Patro, Enigmonia (Anomiidae); Placunanomia, placuna (Placunidae, Fam. Nov.) | |
| Philosophical Transactions of the Royal Society of London B, 276: 453-527. | |
| Young A L (1980) Larval and post-larval development of the windowpane shell, Placuna placenta |
|
| Linnaeus (Bivalvia: Placunidae) with a discussion on its natural settlement. Veliger, 23: 141-148. | |
Web resources
http://www.data.acnatsci.org/obis/ Indo-Pacific Molluscan Species Database at the Academy of Natural Sciences
http://www.seafdec.org.ph/stories/story06-stock-enhancement-program.html provides information on the stock enhancement program. http://www.bfar.da.gov.ph/legislation/fao/fao157.htm describes legislation for conservation of Kapis in the Philippines.